Mitochondria are key to both energy metabolisms in eukaryotic cells and signal transduction of cellular activities. Mitochondrial dysfunction is associated with a variety of human diseases, including mitochondrial encephalomyopathies, cardiovascular diseases, neurodegenerative diseases, and metabolic diseases mainly affecting tissues and organs with high energy demands such as brain, heart, and skeletal muscle. These clinically heterogeneous disorders are collectively known as mitochondrial diseases, with a population prevalence of approximately 1 in 5,000.
Hypertrophic cardiomyopathy (HCM) is the most prominent cause of sudden cardiac death in young individuals. Due to heterogeneity in the clinical manifestations, conventional HCM drugs have limitations for mitochondrial hypertrophic cardiomyopathy. Discovering more effective compounds would be of substantial benefit for further elucidating the pathogenic mechanisms of HCM and treating patients with this condition.
On May 18, 2023, Professor Ruhong Zhou from Shanghai Institute of Advanced Study, Zhejiang University, together with his collaborators published the paper '1-Deoxynojirimycin promotes cardiac function and rescues mitochondrial cristae in mitochondrial hypertrophic cardiomyopathy' in The Journal of Clinical Investigation. They demonstrated the efficacy of 1-Deoxynojirimycin(DNJ) in promoting cardiac mitochondrial function and alleviating cardiac hypertrophy in vivo, and illustrated how DNJ contribute to mitochondria repair and cell function. The work elucidated the mechanism of hypertrophic cardiomyopathy(HCM) and provided a potential therapeutic strategy for mitochondrial diseases.
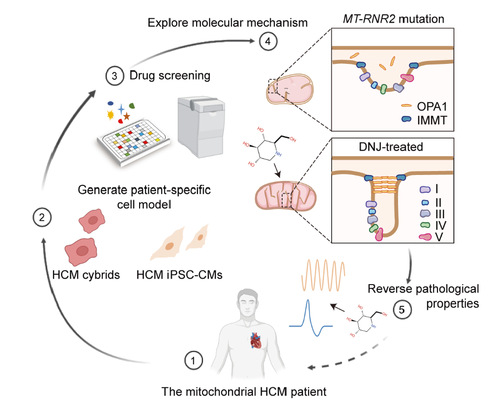
It is previously reported the MT-RNR2 variant associated with HCM that results in mitochondrial dysfunction. Here, the authors screened a mitochondria-associated compound library by quantifying the mitochondrial membrane potential of HCM cybrids and the survival rate of HCM induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs) in galactose media. They developed a two-step drug screening platform by utilizing two cell models, HCM cybrids and HCM PSC-CMs, and results showed that DNJ could rescue abnormal electrophysiological properties and calcium handling, and alleviate physiological defect in HCM iPSC-CMs.
DNJ was further identified to rescue mitochondrial function by targeting optic atrophy protein 1 (OPA1) to promote its oligomerization, leading to reconstruction of the mitochondrial cristae. The authors conducted pulldown-immunoblot verification and concluded that DNJ augmented the level of OPA1 oligomers and improved their biomolecular function. Results showed that DNJ protects the mitochondrial cristae morphology in HCM cybrids, and suggested that DNJ was effective in increasing OPA1 oligomer levels and rescuing mitochondrial function in HCM iPSC-CMs.
The authors further validated the efficacy of DNJ against pathological myocardial hypertrophy in vivo and found that DNJ could improve mitochondrial bioenergetics in cardiac tissue and attenuate AngII-induced cardiac hypertrophy in mice. They further examined the mitochondrial ultrastructure and bioenergetics of cardiac tissues of mice and discovered that AngII-treated mice presented mitochondrial dysfunction and abnormal mitochondrial morphology compared with control mice, while DNJ treatment markedly decreased AngII-induced cristae disorganization and restored RSC stability, MMP levels and ATP production with augmented OPA1 oligomer levels. In contrast, the Sham+DNJ mice group exhibited few changes. Furthermore, abnormalities in the other organs (liver, stomach, colon, kidney, spleen) were not observed in the DNJ-treated groups. These results suggested that DNJ can attenuate AngII-induced cardiac hypertrophy by improving mitochondrial function in mice.
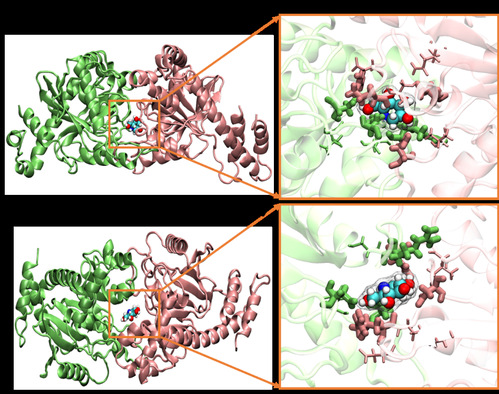
Ligand DNJ promotes OPA1 dimerization
In summary, DNJ was identified for its potential to promote the mitochondrial health and reversed the pathological phenotype of mitochondrial hypertrophic cardiomyopathy. DNJ treatment further recovered the physiological properties of HCM iPSC-CMs by improving Ca2+ homeostasis and electrophysiological properties. An angiotensin II-induced cardiac hypertrophy mouse model verified the efficacy of DNJ in promoting cardiac mitochondrial function and alleviating cardiac hypertrophy in vivo. These results demonstrated that DNJ could be a potential mitochondrial rescue agent for mitochondrial hypertrophic cardiomyopathy, and a convenient preclinical platform for personalized treatment in mitochondrial diseases. To access the work please visit https://www.jci.org/articles/view/164660/pdf.
About Professor Ruhong Zhou
Ruhong Zhou, AAAS Fellow, APS Fellow, is currently Qiushi Chair Professor, Dean, College of Life Sciences; Dean, Shanghai Institute for Advanced Study; and Director, Institute of Quantitative Biology of Zhejiang University. He is also an Adjunct Professor at Department of Chemistry, Columbia University. Before that, he was a Distinguished Research Staff Scientist, and Head of the Soft Matter Science Department at IBM Research.
Professor Zhou’s research focus on Quantitative Biology; Machine Learning, Deep Learning in Biology; Biophysics; and Bio-Nano Interface (nanomedicine). He has authored and co-authored more than 300 journal publications with 25000+ total citations (Google H-index 81), filed 32 international patents and delivered 300+ invited talks at major conferences and universities worldwide. He is part of the IBM BlueGene team that won the 2009 National Medal on Technology (presented by President Obama). He has won the IBM Outstanding Technical Achievement Award (the highest technical award within IBM; 10 times), the IBM Outstanding Innovation Award (twice), and many IBM Research Division Awards. He also won the American Chemical Society DEC Award on Computational Chemistry. He was elected to AAAS Fellow (American Association of Advancement of Science) and APS Fellow (American Physical Society) in 2011. He received his PhD in Biophysical Chemistry from Columbia University, MS in Condensed Matter Physics and BS in Physics both from Zhejiang University.
About SIAS
Shanghai Institute for Advanced Study of Zhejiang University (SIAS) is a jointly launched new institution of research and development by Shanghai Municipal Government and Zhejiang University in June, 2020. The platform represents an intersection of technology and economic development, serving as a market leading trail blazer to cultivate a novel community for innovation amongst enterprises.
SIAS is seeking top talents working on the frontiers of computational sciences who can envision and actualize a research program that will bring out new solutions to areas include, but not limited to, Artificial Intelligence, Computational Biology, Computational Engineering and Fintech.

